The basal level of gene expression associated with chromatin loosening shapes Waddington landscapes and controls cell differentiation
Résumé
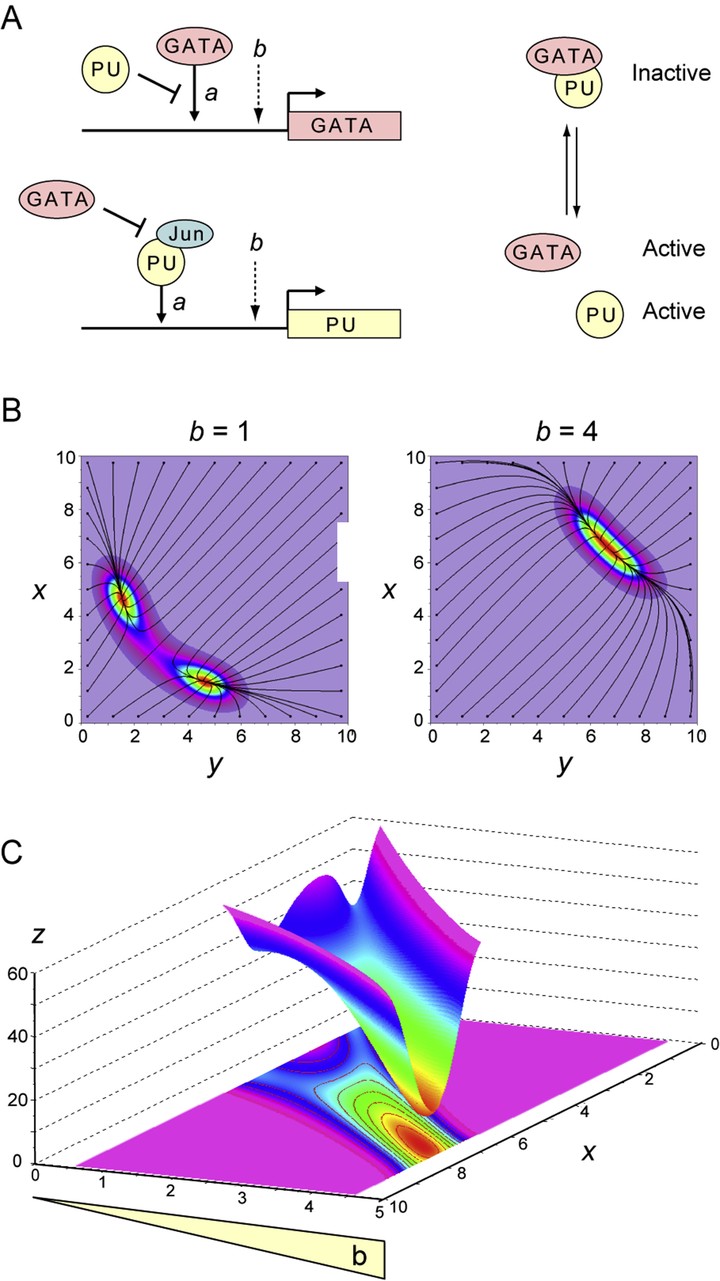
The baseline level of transcription, which is variable and difficult to quantify, seriously complicates the normalization of comparative transcriptomic data, but its biological importance remains unappreciated. We show that this currently neglected ingredient is essential for controlling gene network multistability and therefore cellular differentiation. Basal expression is correlated to the degree of chromatin loosening measured by DNA accessibility and systematically leads to cellular dedifferentiation as assessed by transcriptomic signatures, irrespective of the molecular and cellular tools used. Modeling gene network motifs formally involved in developmental bifurcations reveals that the epigenetic landscapes of Waddington are restructured by the level of nonspecific expression, such that the attractors of progenitor and differentiated cells can be mutually exclusive. This mechanism is universal and holds beyond the particular nature of the genes involved, provided the multistable circuits are correctly described with autonomous basal expression. These results explain the relationships long established between gene expression noise, chromatin decondensation and cellular dedifferentiation, and highlight how heterochromatin maintenance is essential for preventing pathological cellular reprogramming, age-related diseases, and cancer.